by Scott Sabreen, president, The Sabreen Group
Thermoplastics are inherently hydrophobic, low surface energy substrates that do not adhere well to other like or dissimilar materials. Semicrystalline polymers are generally more difficult to bond (compared to amorphous) due to their ordered structure morphology, e.g., polyolefins, polyamides (nylons), acetals, polyesters, polyphenylene sulfide, etc.
While factors such as surface texture topography improve wetting, the use of surface modification solves most bonding problems. Common surface modification methods include electrical (corona) discharge, flame plasma, atmospheric plasma, cold-gas plasma and UV radiation-ozone. Surface oxidation pretreatment of polymeric materials introduces polar reactive groups, which improve surface free energy and, consequently, the wettability and bondability of these surfaces. While each of these processes generates highly reactive gas plasmas, it is important to recognize there can be significant differences between treated surfaces.
This article discusses flame treatment of three-dimensional plastic products as a valuable technique for improving the surface properties of polymers – starting with combustion process principles followed by special focus on laminar flame zones, burner selection and flame stability, zero gas pressure regulators, treatment life and process control factors. The independent information is presented in hopes of reducing misnomers regarding flame treatment and promoting its effectiveness when compared with alternative pretreatments.
Flame plasma pretreatment
Flame treatment is a gas-phase pretreatment method in which the combustion of a hydrocarbon fuel, under controlled conditions, generates the flame plasma that modifies the substrate surface without affecting the bulk properties of the polymer. The oxidation of the polymer surface as a result of flame treatment occurs through multiple physical processes, including homogeneous combustion of the premixed reactants, transport of the products of combustion to the polymer surface and heterogeneous reaction of flame products with the polymer. The adiabatic flame temperature is approximately 3,300°F (1,816°C). The mechanism of flame treatment is the controlled combustion of a hydrocarbon fuel to generate the flame plasma. Premixed laminar flames produce an exothermic reaction. The exact ratio of oxidizer to fuel needed for complete combustion is known as the stoichiometric ratio. At stoichiometric combustion there is no excess oxygen or fuel. The stoichiometric ratio is approximately 10:1 for natural gas and 24:1 for propane.
The flame is a mixture of a fuel and an oxidizer thoroughly mixed before combustion. The molar ratio of the fuel to the oxidizer is probably the most important parameter within the flame treatment process. For natural gas, the following equation describes the combustion reaction:
CH4 + 2O2 + 8N2 → CO2 + 2H20 + 8N2 + flame plasma
The three main process control variables are flame chemistry, distance of the substrate from the flame and dwell time of treatment. In a combustion system the flame is a subsonic wave characterized by a velocity called laminar flame speed, which is defined as the velocity at which unburned gases move throughout the combustion wave in the direction normal to the wave surface. A laminar flame profile consists of three zones – reducing, luminous and post-combustion. Each zone has different temperature gradients and chemical reactive species. Placement of the part moving through the flame is critical and must be precise for maximum surface wetting and functionalization.
Optimal treatment (oxidation) occurs in the main reaction luminous zone or oxidizing. This is the hottest flame region, where temperature of the combustion system reaches approximately 1,700°C for natural gas and 1,900 to 2,000°C for propane-based mixtures. In this zone, free radical reactive species content (including hydroxyl, carbonyl, carboxyl, ether and ester) increases dramatically to the detriment of the reactant concentration. The high concentration of radical species makes this region strongly oxidizing, in contrast to the reducing zone.
The color of this zone depends on the fuel/air ratio. A deep bluish violet radiation, with the flame becoming almost transparent if the quantity of gas is increasingly reduced, is produced when the mixture is gas-lean (due to excited CH radicals). A green radiation appears when the mixture is gas-rich (due to excited C2 molecules). When the gas in the mixture increases still further, the radiation turns yellowish because of the carbon particles formed. Ideal flame chemistry is that which provides for an oxygen concentration in the flame plasma that is, after the combustion reaction, of 0.1% to 0.5%.
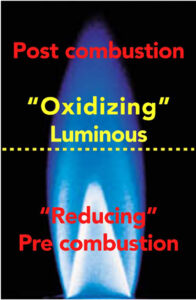
The optimal treatment distance (position) of the substrate from the flame (just above the pre-combustion reducing zone) is typically between 3/8″ and ½” (9.5 mm to 12.7 mm). The actual treating portion of the flame extends approximately 1½” (38.1 mm) beyond the flame tip, with about ½” (12.7 mm) producing the highest level of treatment. Reference the yellow dotted line in Diagram 1. The surface to be treated should never contact the reducing zone (sub-stoichiometric) of the flame.
The pre-combustion or reducing zone is the coldest region in the flame, where the premixed unburned gases have not reached the optimal oxidizing condition. Characterized by its bright steel-blue color, this region is ineffective for surface activation and offers no oxidation benefits. The post-combustion zone is the largest of the three regions in a laminar flame profile. The temperature remains high due to the exothermic oxidation reaction of CO into CO2, with a release of heat.
This region is characterized as intermediate, between reducing and luminous zones, in terms of temperature and oxygen radicals concentration. The high temperature burned gases generally present a reddish color, given by water vapor and carbon dioxide (combustion products with hydrocarbon combustibles) radiation and some dirt particles.

The gap (distance) between the luminous flame and substrate surface is a critical factor in determining the extent of activation accomplished by the treatment. Generally, when the substrate passes through the flame, a rapid depletion in the wettability of the treated surface occurs as the distance increases between the cones of the flame and substrate. However, a beneficial effect arising from the treatment is still appreciable several millimeters beyond the post-combustion zone. To maximize the benefit from the treatment, the flame should work in tandem with its luminous zone, which is the richest in active oxidizing species (OH radicals and O atoms) and the one at the highest temperature within the whole combustion system3.
Since a greater extent of oxidation is concentrated near the outermost shallow surface region (5 to 10 nm), flame-treated surfaces often result in improved wettability and retain more stable aging (shelf life) than corona-treated surfaces. Flamed polymers also may demonstrate improved wetting properties due to a different mix of chemical functional groups. Different oxidized functionalities will make varying contributions to the wettability of a surface-oxidized polymer.
Studies demonstrated that, while both corona-treated and flame-treated polyolefins are surface oxidation processes, the two methods differ considerably in the mechanism of oxidation.

The extensive scission associated with corona processes affects the wettability and stability of the treated polypropylene (PP) films. By contrast, flame surface oxidations are dominated by OH radical reactions that lead to oxidation without low molecular weight oxidized materials (LMWOM) formation. The more limited chain scission associated with flame treatment leads to the formation of topographic nodules of intermediate molecular weight materials that are much smaller than the mounds of LMWOM. Because of the lack of LMWOM formation, flame-treated PP is more highly oxidized after washing with water, more wettable and more stable than corona-treated PP.
Treatment stability decreases in the extent of oxidation or losses in wettability as a function of aging are usually attributed to the reorientation or localized migration of oxidized functional groups located near the surface of a modified PP. As the molecular weight of the surface region decreases, the tendency for functional-group reorientation or migration should increase. The molecular weight of a corona-treated polypropylene surface is lower than that of a flame-treated PP surface. Therefore, corona-treated PP is less stable than flame-treated PP. Treatment stability is an example of how the surface molecular weight can be more important than the extent of oxidation in determining a practical performance property1.
Zero gas pressure regulators should be installed with premix burner systems, using venturi air/fuel mixers to maintain a constant air/fuel ratio regardless of the burner firing rate. The venturi mixer is a proportional mixing device that has inlets for both the fuel gas (natural or propane) and air/oxygen, and inside the body mixes these gases properly and distributes them through the outlet. Functionally, as combustion air flows through the orifice in the air/fuel mixer, it causes a pressure drop that is sensed internally as a negative pressure through a downstream sensing tube in the zero gas pressure regulator – to maintain zero pressure relative to atmosphere.
When the negative pressure is sensed, the zero gas pressure regulator will open enough so that the positive pressure equals the negative suction pressure. As the combustion airflow increases or decreases, based on the energy requirements of the process, the suction will increase or decrease accordingly, and the zero gas pressure regulator will open (with increasing combustion airflow) or close (with decreasing combustion airflow) to always maintain zero pressure. In this way, no matter what the combustion airflow is, the air/fuel ratio should stay relatively constant.
Most zero gas pressure regulators are designed not to be adjusted, since the regulator spring is designed to precisely balance the weight of the valve internals. There are newer models available that allow for adjustment of the balancing spring. This provides added flexibility. When selecting a zero gas pressure regulator consider turndown – maximum to minimum fuel flow. More is better.
Also consider the size of the diaphragm. Larger is better since it provides more precise control. In sizing zero gas pressure regulators, keep in mind that the pressure drop across the regulator is equal to the inlet pressure, since the outlet pressure is zero.
Proper selection of burner type is critical to the uniformity and totality of treatment. Product design, geometry and polymer substrate are important factors. A well-designed burner will have a high level of stability, reducing the occurrence of flashback and flame lift, which are two types of instabilities that occur due to an imbalance between the fuel/oxidizer mixture flow velocity and the burn velocity of the combustible mixture. Flashback can develop in a premixed burner if the burning velocity of the flame exceeds the stream velocity flowing out of the port. Under these conditions, the maximum port diameter ensuring necessary thermal losses has been exceeded, thus allowing the flame to propagate back into the mixing chamber.

Two types of burners are used for flame treating: ribbon and drilled port. Ribbon burners consist of a series of crimped stainless steel ribbons inserted into brass, stainless steel, mild steel and cast iron. The number of ribbons, the crimp pattern and number of burner ports and other ribbon pattern design features depend on the application. Ribbon burners can offer advantages because of their ability to provide large flame surface and flame stabilization. The second type of burner is the drilled port. Drilled port burners are normally manufactured in stainless steel and aluminum.
Ribbon burners are the latest generation of burners and most widely adopted solution at an industrial level because the flame patterns are customizable by adjusting the width of the slot and configuring the ribbons3. Ribbon burners are the optimal solution for the surface modification or flame treatment of polymer films2.
The ribbon comprises packed sheets of corrugated stainless steel that form rows of nearly elliptical outlets, or ports, each having a major diameter of 2.4 mm and a minor diameter of 1.5 mm. The rows are parallel but offset so that the centers of the outlets are staggered. Single slot and triple slot burners are widely utilized, along with other designs, including internal firing ring burners.
Dwell time is an important process factor in optimizing the surface treatment chemical functionality. The part must be in contact with the flame plasma for sufficient time for the reaction kinetics to be maximized. Brief contact time is controlled by how fast the part is passed through the flame and is dependent on burner width, plasma output and type of resin. For example, polypropylenes may require different dwell time than polyethylenes. Optimum treatment requires a spatially homogeneous post-flame reaction zone, even with burners up to three meters in length.
Temperature and relative humidity of the surrounding environment are important. Increases in either can cause a shift in the gas/air mixture toward a fuel-rich composition, thereby triggering a change in the properties of the treated polymer surface. Careful monitoring is beneficial for robust results. As with all plasma pretreatments, over-treatment must be avoided, as it can lead to artificially high surface energy and adhesion or delamination field failures. Under-treatment, due to improper flame set-up or combustion system design, also must be avoided.
Flame treatment of 3D polymer products is almost always conducted using a single pass, in contrast to plastic films that are often treated two times by web. Theoretically, a rapid single-pass exposure of the product through the high temperature flame will not trigger the migration of low-molecular weight additives (plasticizers, heat/UV stabilizers, release agents, antistatics, etc.) to the surface, inhibiting adhesion.
In addition to surface modification benefits, flame treatment adds value to plastics molding operations. For example, injection-molded “top gate” polyolefin closures have a small nub in the middle of the closure (exterior top surface) due to the pinpoint gate location. Flaming smooths this surface defect (due to heat) and removes contaminants for aesthetic printing quality. Compression molded closures do not have this issue. Thus, surface pretreatments may offer multiple benefits in specific applications that need to be considered when procuring equipment systems.
Conclusion
Flame treatment is an excellent process for improving polymer adhesion by increasing surface energy and chemical functionality. It is well suited for treating large surface areas that have contours and/or recesses. Fully automated robotic systems are highly effective and easily integrated.
Unlike other atmospheric pretreatment methods, flaming removes most surface debris and some hydrocarbon contaminants. Burner selection and zero gas pressure regulators are critical components. It is essential to understand how to control the process variables to maintain robust flame treatment since minimal changes can cause significant deviations in the expected results and subsequent adhesion bonding.
All flame treatment equipment is not created equal. Carefully evaluate all components and combustion system design. It is important to state that although generally valid, the concepts outlined in this paper may not apply in every application. Some aspects may need to be individually examined, e.g., specific substrate type, gas fuel type, distance and dwell time.
References
- A Comparison of Corona-Treated and Flame-Treated Polypropylene Films, M. Strobel et al. 2003
- Application of Ribbon Burners to the Flame Treatment of Polypropylene Films, Alexander, Branch, Strobel 2008
- The fundamentals of flame treatment for the surface activation of polyolefin polymers – A review, Farris et al. 2010
- Flame Plasma Surface Treatment Improves Adhesion of Polymers, Joseph DiGiacomo, Scott Sabreen, Plastics Decorating Magazine, 2005
- Flynn Burner Equipment Manual, Surface Treatment Air Gas Mix System with Flame Safety Control
 Scott R. Sabreen is founder and president of The Sabreen Group, Inc., an engineering consulting company specializing in secondary plastics manufacturing processes – surface pretreatments, adhesion bonding, inkjet printing, laser marking, decorating and finishing and product security. Sabreen has been developing pioneering technologies and solving manufacturing problems for more than 30 years. He can be contacted at 972.820.6777 or by visiting www.sabreen.com.
Scott R. Sabreen is founder and president of The Sabreen Group, Inc., an engineering consulting company specializing in secondary plastics manufacturing processes – surface pretreatments, adhesion bonding, inkjet printing, laser marking, decorating and finishing and product security. Sabreen has been developing pioneering technologies and solving manufacturing problems for more than 30 years. He can be contacted at 972.820.6777 or by visiting www.sabreen.com.


