by Scott R. Sabreen, president, The Sabreen Group
Many polymers are inherently hydrophobic, low-surface energy substrates that do not adhere well to like or dissimilar materials. Surface modification methods are proven to solve most adhesion problems. Traditional methods include electrical (corona) discharge, flame plasma, atmospheric plasma, cold-gas plasma and UV radiation-ozone. Surface oxidation pretreatment of polymeric surfaces introduces polar reactive groups that improve surface-free energy and consequently the wettability and bondability of these surfaces. A newer method, PYROSIL® flame treatment, hereafter referred to as process or treatment, is rapidly emerging as an effective process for improving adhesion on tough-to-adhere polymers.
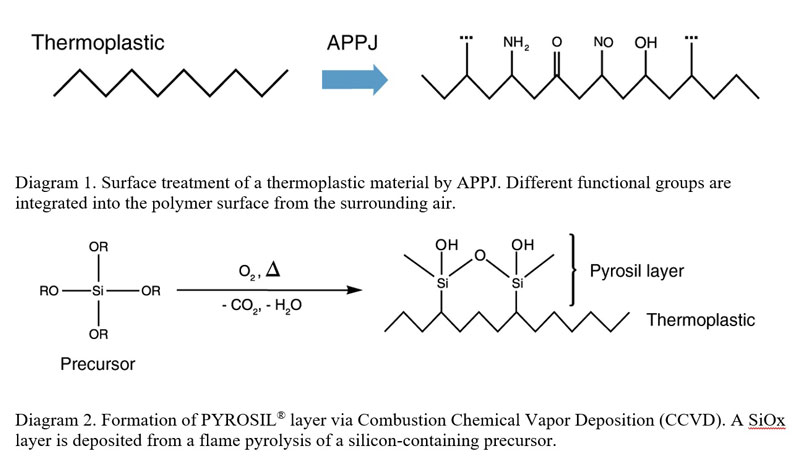
This treatment is a surface modification technique that deposits an amorphous silicon dioxide (SiO2) layer on a substrate surface via combustion chemical vapor deposition (CCVD). The equipment and processes were developed in the 1980s, leading to the first successful commercialization of the technology used to increase bonding with adhesives. By means of flame pyrolysis, a thin layer (5-100 nm) of highly reactive and hydrophobic silicone oxide can be achieved on the substrate. Consequently, a defined hydrophilic surface is built due to the high density of Si-OH groups on the surface of the deposited SiO2 particles and nanoscale roughness. Silicon-containing films find many applications for polymers, glass, ceramics and metals.
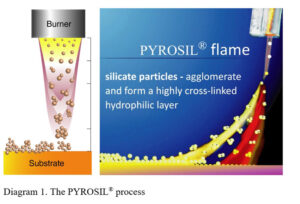
In the process, the product to be treated is fed through a laminar gas flame, doped with a silicon-containing precursor material, in a manner similar to traditional flame treatment (Figure 1). Due to the brief flame-substrate interaction, the process can be used on temperature-sensitive plastics materials.
CCVD is a chemical process by which thin-film coatings are deposited onto substrates in the open atmosphere. In the CCVD process, a precursor compound is added to the burning gas. The flame is moved closely above the surface to be coated. The high energy within the flame converts the precursors into highly reactive intermediates that readily react with the substrate, forming a firmly adhering deposit. The microstructure and thickness of the deposited layer are controlled by process parameters including speed of substrate or flame, number of passes, substrate temperature and distance between flame and substrate. Silicon dioxide layers are the most commonly deposited layers. Freshly deposited layers are highly reactive and can serve as adhesion-promoting layers for polymer coatings and bonding.
Compared with flame treatment
This process and traditional flame plasma treatment have process similarities, but the flame chemistry and surface functionality mechanisms are different1.

Both methods are gas-phase surface oxidation processes in which the combustion of a hydrocarbon fuel, under controlled conditions, generates the flame plasma that modifies the substrate surface. Premixed laminar flames produce an exothermic reaction. The treatment flame is pinkish in color, while the traditional flame treatment is bluish in color (Figure 2).
The main difference is that this process produces SiO2 and flame treatment does not. Consequently, a defined hydrophilic surface is built due to the high density of Si-OH groups on the surface of the deposited SiO2 particles. Similar combustion equipment, ribbon burners and process set-up/control are utilized. The three main process control variables are flame chemistry, distance of the substrate from the flame and dwell time of treatment. Treatment must take place in the main reaction luminous zone or oxidizing1.
For this treatment, one equation for the combustion of organosilicon compound tetramethysilane “TMS” and the formation of SiO2 is:
Si(CH3)4 + 13 O2 + C3H8 → SiO2 + 10 H2O + 7 CO2
For natural gas flame treatment, the following equation describes the combustion reaction:
CH4 + 2O2 + 8N2 → CO2 2H2O + 8N2
Both combustion reactions produce CO2 and H2O water byproducts.
Compared with cold-gas plasma
Prior to the invention of this process, one of the most practiced methods for SiO2 thin-film deposition is plasma-enhanced chemical vapor deposition (PECVD), which is performed in remote chambers under vacuum low-pressure glow discharge plasma. As the name implies, the process is a chemical vapor deposition vs. CCVD, a combustion vapor deposition.
PECVD is conducted at lower temperature than standard chemical vapor deposition and is more versatile in producing many different types of film deposition coatings and functionalized surfaces.
The major drawbacks are offline batch operations and high equipment/consumable costs. This process is conducted at ambient conditions, ideal for cost effective inline operations2.
Research study: Bonding of low surface energy thermoplastics
There are extensive research studies documenting this process. One study conducted by V. Seitz et. al compares atmospheric pressure plasma jet (APPJ) and a PYROSIL® flame for improved bonded strength on thermoplastics polybutylene terephthalate (PBT), polyetheretherketone (PEEK) and polyphenylene sulfide (PPS)3. This study explains lower contact angles were achieved using the treatment and verified with photoelectron spectroscopy (XPS), 90°– peel test data, and surface analysis.
Chemical composition of treated surfaces
See Diagrams 1 and 2.
Contact angle (CA) measurement
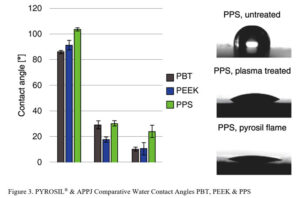
Water contact angles were measured on reference samples and after different surface treatments (Figure 3). The untreated thermoplastics exhibited a mainly hydrophobic behavior, as measured contact angles ranged between 86.2° (PBT) and 104.0° (PPS).
Plasma treatment, as well as the process flame, had a significant influence on the wetting behavior of all tested thermoplastics leading to a clear decrease of the contact angle. For PBT, the contact angle decreased to 29.0° (plasma) and 10.1° (the new process). The hydrophilization of PEEK surfaces (reference 91.6°) was even higher, resulting in water contact angles of 17.7° (plasma) and 10.5° (new process).
For PPS, which offers the highest contact angles of the tested polymers, contact angles of 30.3° after atmospheric plasma treatment and 24.0° after the flame treatment were measured. The contact angle measurement confirms a hydrophilization of the thermoplastic surfaces by APPJ and the treatment flame as a significant decrease in water contact angle occurs. This improvement in wetting behavior is induced by the formation of functional groups on the polymer surface.
The plasma treatment utilizes ambient air as a process gas, so oxygen and nitrogen containing groups are created on the surface. These polar groups improve the wetting behavior by water and the contact angle decreases. The process flame produces a thin SiOx-layer of some nm thickness on the polymer surface.
The wetting behavior of this layer with contact angles between 10.1° (PBT) and 24.0° (PPS) indicates the formation of a high amount of hydrophilic silanol groups, which have a high polarity compared to the nonpolar thermoplastics.
The XPS survey spectrum of the process-treated PEEK samples shows appearance of the Si2s and Si2p peaks, which is evidence for the deposition of a silica-containing layer on the thermoplastic surface, as untreated samples do not show these peaks.
On PPS surfaces, a clear increase in Si can be observed on the treatment samples compared to the reference sample. Moreover, a remarkable increase of oxygen can be observed on both PEEK and PPS treated surfaces, which is also a result of SiOx deposition.
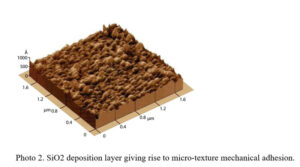
Atomic force microscopy (AFM, Figure 4) shows an SiO2 deposition layer, which increases mechanical adhesion via micro-texture4.
Conclusion
This treatment is a process for improving adhesion on polymers, glass and metals and is suited for treating large, molded surface areas with contours and flat blown films. Automated systems are highly effective and easily integrated. Unlike other gas-phase surface oxidation methods, this treatment (CCVD) is a chemical process by which thin-film SiO2 coatings are deposited onto substrates in the open atmosphere. PYROSIL® and traditional flame treatment have process similarities, but the flame chemistry and surface functionality mechanism are different. Combustion system design, including all components, is critical for optimal treatment. Minimal set-up changes can result in adhesion bonding results. It is important to state that, although generally valid, the concepts outlined in this paper may not apply in every application. Some aspects may require individual examination.
References
- Sabreen, S. Flame plasma surface modification of polymers for adhesion bonding: process control, equipment and applications. Plastics Decorating magazine July/August 2020.
- Sabreen, S. Cold Gas Plasma Surface Modification – Optimize Plastics Bonding Adhesion. Plastics Decorating magazine January/February 2010.
- Seitz V., K. Arzt, Mahnel S., Rapp C., Schwaminger S., Hoffstetter M., Wintermantel E., (2015). Improvement of adhesion strength of self-adhesive silicone rubber on thermoplastic substrates – comparison of an atmospheric pressure plasma jet and a PYROSIL® flame. International Journal of Adhesion & Adhesives, Volume 66.
- AST/SURA Instruments PYROSIL® Brochure. A technology with a broad range of applications, 2004.
 Scott R. Sabreen is founder and president of The Sabreen Group, Inc., an engineering consulting company specializing in secondary plastics manufacturing processes – surface pretreatments, adhesion bonding, inkjet printing, laser marking, decorating and finishing and product security. Sabreen has been developing pioneering technologies and solving manufacturing problems for more than 30 years. He can be contacted at 972.820.6777 or by visiting www.sabreen.com.
Scott R. Sabreen is founder and president of The Sabreen Group, Inc., an engineering consulting company specializing in secondary plastics manufacturing processes – surface pretreatments, adhesion bonding, inkjet printing, laser marking, decorating and finishing and product security. Sabreen has been developing pioneering technologies and solving manufacturing problems for more than 30 years. He can be contacted at 972.820.6777 or by visiting www.sabreen.com.

